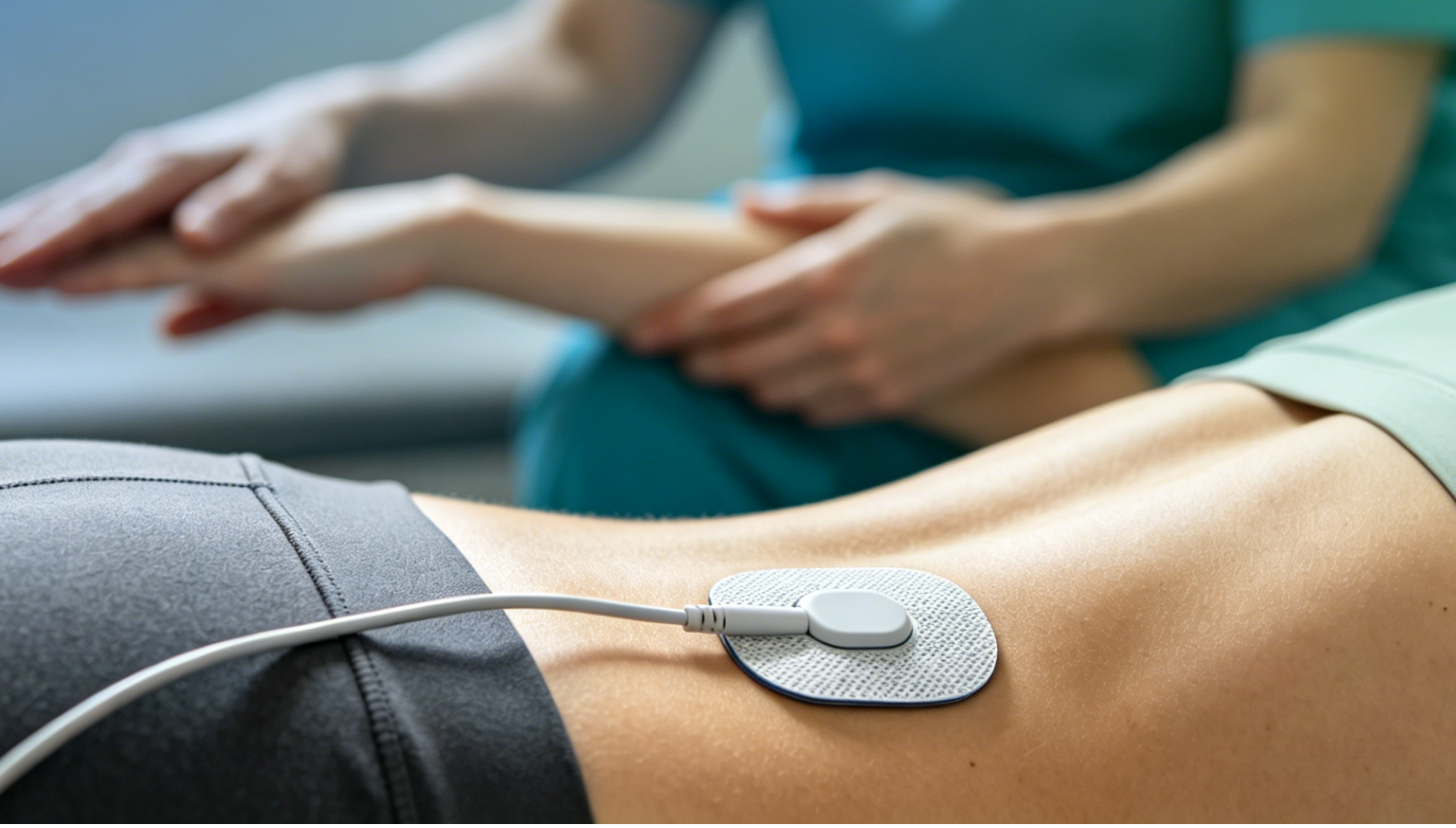
Among the spectrum of chronic pain disorders, fibromyalgia syndrome is undoubtedly one of the most perplexing and distressing. Patients are neither malingering nor suffering from a purely psychological condition; rather, their nervous systems behave like a radio whose volume has been turned up to maximum, distorting normal tactile and pressure signals into sharp, pervasive pain. Widespread body pain, profound fatigue, and cognitive dysfunction (“fibro fog”)—these symptoms act as invisible shackles, severely eroding quality of life. Worse still, conventional analgesics often yield little benefit. However, a landmark clinical study published in 2026—the FM‑TIPS trial—has kindled a beacon of hope by integrating technology with rehabilitation for this long‑neglected population.
The Treatment Dilemma: Exercise as a “Double‑Edged Sword”
For most musculoskeletal conditions, exercise is medicine. But for fibromyalgia patients, exercise represents a formidable barrier. The core obstacle they face is termed exercise‑induced pain. Whereas a healthy individual might experience mild muscle soreness after physical activity, a fibromyalgia patient may endure an intense pain flare lasting hours or even days after gentle stretching or a short walk. This fear instinctively drives patients to avoid activity, leading to physical deconditioning, muscle wasting, social isolation, and a vicious downward spiral. Clinicians have long sought a method that encourages movement without inflicting a disproportionate punishment of pain.
The Striking Findings of the FM‑TIPS Study: Adding a “Shock Absorber” to Exercise
The FM‑TIPS (Fibromyalgia TENS in Physical Therapy Study) trial, led by a team including Professor Kathleen Sluka of the University of Iowa, was a rigorously designed real‑world study. Researchers divided fibromyalgia patients into two groups: one received four weeks of standard physical therapy (including gentle stretching and aerobic conditioning), while the other received the same physical therapy with the concurrent application of TENS over painful areas during each session.
The results were clinically significant and have begun to shift existing treatment guidelines:
- Significant Reduction in Movement‑Evoked Pain: Patients in the TENS combined treatment group reported markedly lower immediate pain levels while performing rehabilitative exercises. TENS effectively acts as a “shock absorber” on the nervous system, rendering the mechanical stimuli of movement far less threatening.
- Durable and Far‑Reaching Effects: This improvement was not transient. The study showed that benefits emerged by day 30 and persisted for at least six months after the intervention concluded. This suggests that TENS combined with movement may help reset the heightened sensitivity of the central nervous system.
- Efficacy Comparable to First‑Line Medications: When the research team compared the effect size of TENS to FDA‑approved first‑line medications for fibromyalgia (such as pregabalin and duloxetine), they found that TENS was comparable or even superior in reducing exercise‑induced pain—and did so without common drug side effects like drowsiness, dizziness, or weight gain.
The Roovjoy Perspective: Empowering Patients to Reclaim Life
This 2026 breakthrough study substantially elevates the role of TENS in managing complex chronic pain. It demonstrates that TENS’s value extends beyond merely “stopping the pain of the moment.” It serves as an enabling tool that helps patients overcome the psychological barrier of “being afraid to move.” When patients hold a user‑friendly, at‑home TENS device—such as those manufactured by Roovjoy Medical—they gain a sense of control. They know that if discomfort arises during rehabilitation, they possess an immediate, drug‑free tool to address it.
For millions of fibromyalgia patients worldwide, TENS combined with physical therapy is no longer merely an alternative approach; it is a preferred strategy grounded in high‑level evidence‑based medicine. It uses a gentle current to leverage the body’s immense capacity for self‑repair, enabling patients to step outside and embrace sunlight and life once again.
Post time: Feb-20-2026